1. What Happened?
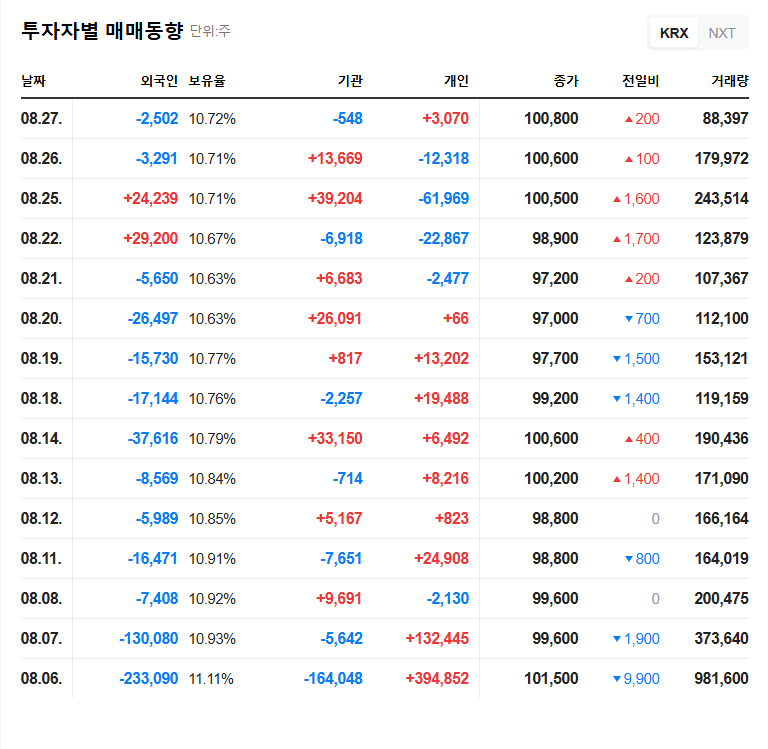
AptaBio has applied for Investigational New Drug (IND) application approval from the US FDA for a Phase 1 clinical trial of its oral macular degeneration treatment, ABF-101. The trial aims to evaluate the safety, tolerability, and pharmacokinetics of ABF-101 in healthy volunteers and patients with moderate to advanced AMD. The company addressed the FDA’s request for supplementary in vitro test data, stating they can respond promptly.
2. Why Does It Matter?
AMD is a leading cause of blindness, and the demand for effective treatments is high. The development of an oral treatment offers significant advantages in patient convenience and has substantial market potential. This IND application is a crucial milestone, demonstrating AptaBio’s drug development capabilities. Positive clinical results could significantly increase the company’s value. Their other pipelines, including the Apta-DC platform (for blood cancer, pancreatic cancer, etc.) and the CAF Modulation platform (for immuno-oncology), are also noteworthy.
3. What Should You Do?
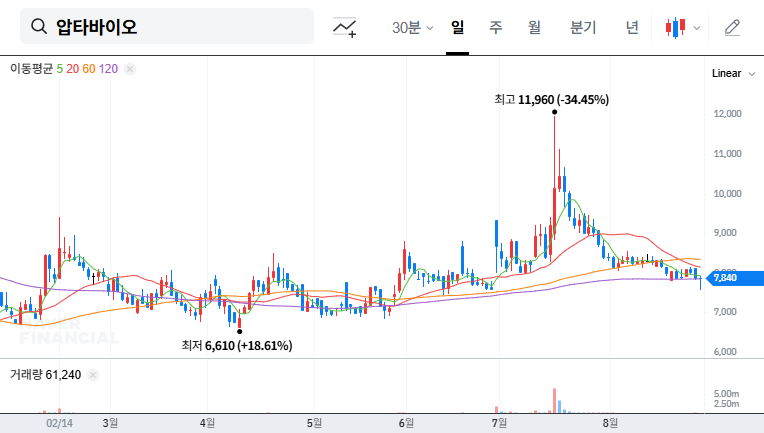
While this is positive news, it’s crucial to consider the inherent uncertainties of drug development. Careful monitoring of Phase 1 results, the success of technology transfer, and AptaBio’s financial health are essential. Focus on a long-term investment strategy rather than short-term stock fluctuations. Given the volatility of the biotech sector, diversification is recommended.
4. Action Plan for Investors
- Step 1: Gather Information – Continuously check for updates on clinical trial progress, technology transfer negotiations, and financial disclosures related to AptaBio.
- Step 2: Analyze and Evaluate – Analyze AptaBio’s investment value based on the information gathered. Consider your investment goals and risk tolerance to decide whether to invest.
- Step 3: Diversify and Monitor – If you decide to invest, diversify your portfolio to mitigate the high volatility of the biotech sector. Continuously monitor your investment to manage risk.
FAQ
What is ABF-101?
ABF-101 is an oral treatment for macular degeneration being developed by AptaBio.
What will the Phase 1 clinical trial evaluate?
It will evaluate safety, tolerability, and pharmacokinetics.
What are the key investment considerations?
Investors should consider the uncertainties of clinical development and the company’s financial health.